PATIENT AND PUBLIC INVOLVEMENT (PPI)
Putting Patients and the Public at the Heart of Our Research
In the Cancer Prevention Trials Unit CPTU, we put patients and members of the public at the heart of everything we do. We are committed to patient-centred research, involving patients and the public at every level - from project design to results dissemination. We have the support of a PPI Coordinator to ensure this involvement is meaningful and sustainable.
Since our trials are typically population-based, patient and public involvement (PPI) plays a crucial role to ensure our participants are diverse and represent of a range of backgrounds, experiences and views. PPI also helps us to ensure that the results of our cancer screening and prevention studies are applicable and meaningful to the target populations. Active and sustained engagement with members of the public and community groups is key to not only ensuring dissemination but also wide uptake of study findings among those who may benefit from screening.
Patient and Public Involvement Across the Research Pathway
This diagram shows the different stages of the research pathway and how patients and the public can contribute at each stage.
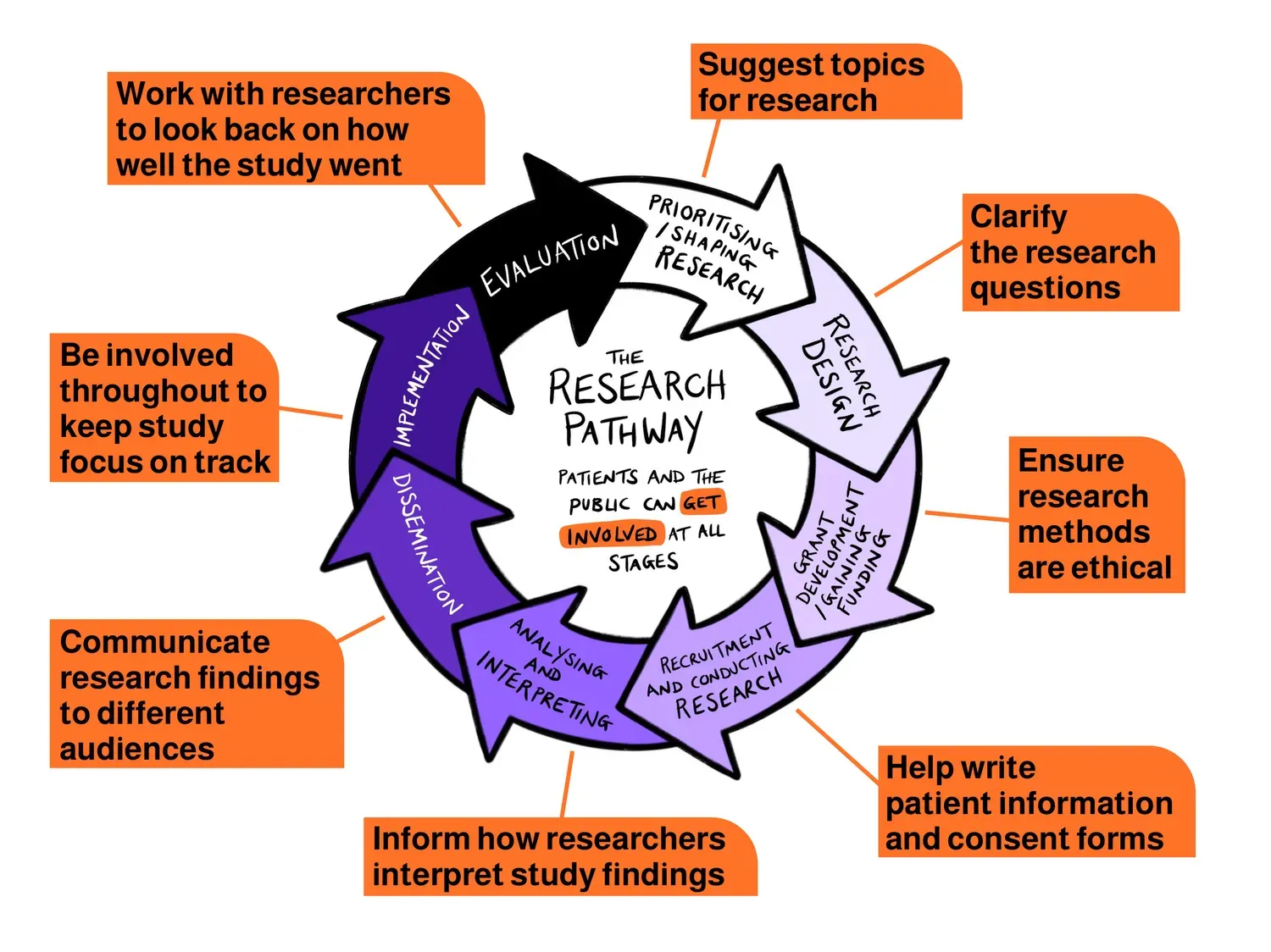
CPTU Patient and Public Involvement Panel:
Our standing panel of patient and public contributors plays a vital role in shaping our unit's activities and research priorities. Panel members are actively involved in key decision-making, and are also part of the Strategic Advisory Board, and Scientific Committees, as well as discrete projects.
Our Patient and Public Involvement pool:
Within our centre we have built a pool of 200 contributors to support ad-hoc PPI activities. This pool helps us bring in diverse perspectives and quickly engage people for new projects and activities.
Want to Get Involved?
If you are interested in getting involved as a patient or public contributor, you can join our Patient and Public Involvement Pool by signing up here.
Case study: PPI and Corrective and Preventive Actions
The Cancer Prevention Trials Unit (CPTU) has developed a new process with our Patient and Public Involvement (PPI) panel to include patient and public perspectives for Corrective and Preventive Action Plans or ‘CAPAs’. These plans are usually written by trials unit staff when deviations occur during trials (when something doesn’t go as planned).
Our CPTU PPI panel piloted the new approach using an example of a past deviation and gave us feedback on the process.
As a result, we will now involve at least one PPI reviewer in CAPAs where a deviation relates to participant data, experience, or a complaint. This will help ensure that the actions taken to address issues and prevent recurrence are informed by, and aligned with, patient and public perspectives.
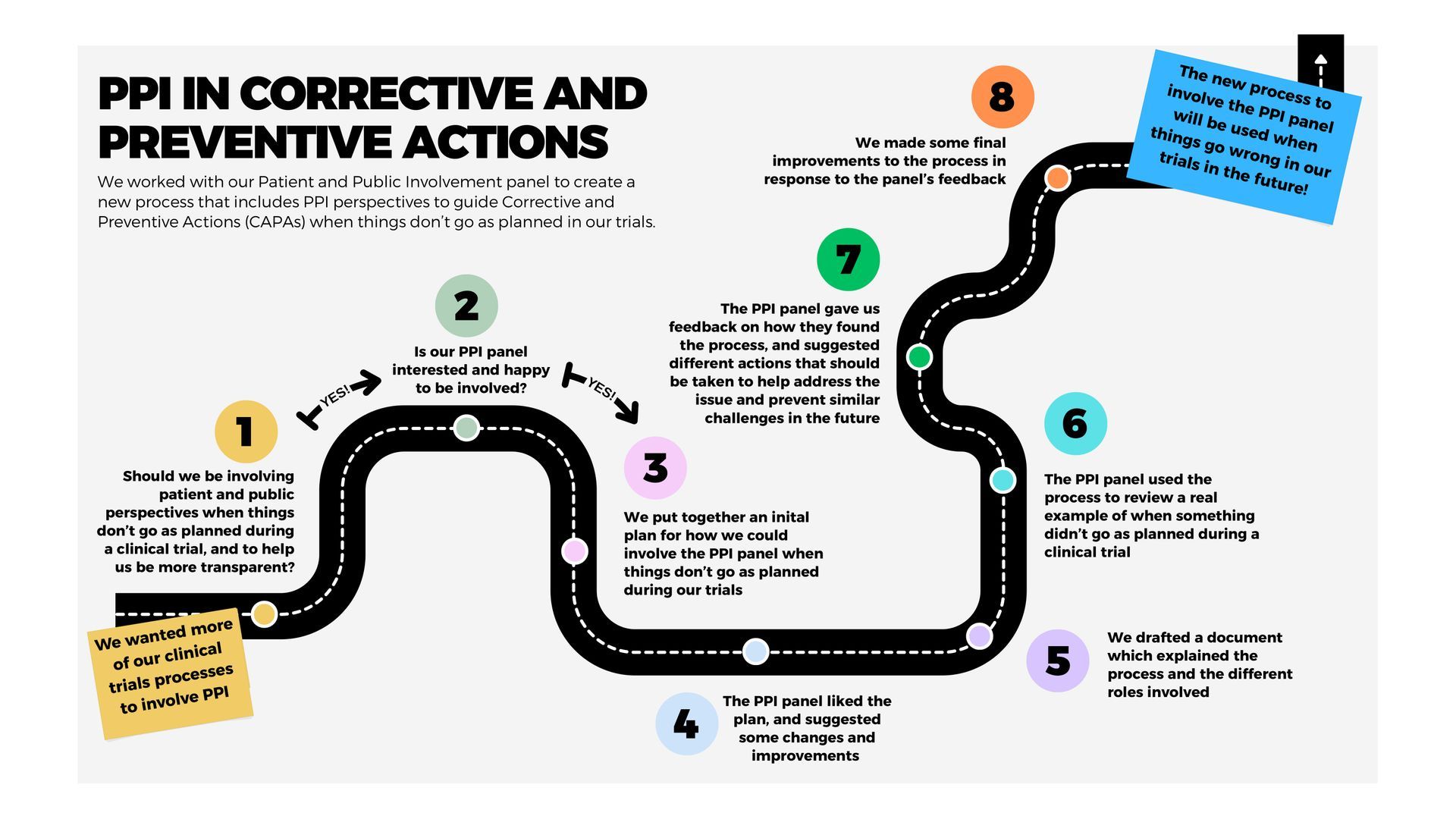
Please see here for a more detailed diagram of the new process.
Jane Cavanagh, CPTU PPI Panel Member, on being involved in the development of the new CAPA process
“Clinical trials reviews often focus on technical analysis and can spend less time focusing on the lived experience of the participants. A PPI perspective can help to improve communication and transparency, supports more ethical and participant-focused responses and can lead to more practical and effective solutions. Involving patients also helps rebuild trust, reduces blind spots in decision-making, and contributes to preventing similar problems in future trials.
I found working with the CPTU team to develop a new process for incorporating Patient and Public perspectives into Corrective and Preventative Action plans to be highly collaborative, interesting and constructive. The team was open to feedback and genuinely committed to ensuring that patient voices were meaningfully embedded rather than treated as an afterthought. Communication was clear throughout, and there was a shared focus on creating a practical and sustainable approach that enhances both the quality and accountability of clinical trial processes.”
Reshma Punjabi, CPTU PPI Panel Member, on being involved in the development of the new CAPA process
“The science of clinical trials is often not well understood in the general population.
Should things not go to plan during clinical trials, it is important that we can communicate with participants why this has happened and that any issues will be addressed with care and rigour. This is key to maintaining participants’ trust and confidence in the trials processes. It is also very important that when communicating with the participants and their carers that this is done in a patient-friendly, reassuring and culturally sensitive manner.
This is why I found working with the CPTU to develop the Corrective and Preventive action plans thought-provoking and fulfilling. I was very pleased at how the PPI panel’s different perspectives, mine being from a patient and carer perspective, were involved to develop the new process to resolve issues which the clinical trials team might have missed, as well as how this is communicated to participants. The process of having the PPI panel working alongside the CPTU staff should be reassuring to the public that the trials are being conducted in a transparent and accountable way.”
Active PPI Across Projects:
For each project, we actively involve patients and members of the public throughout the research lifecycle from grant writing and research design to the results dissemination phase. We also partner with community organisations, particularly those supporting under-served groups, to ensure we can learn from their experiences and perspectives and share study findings directly with relevant participant groups.
Impactful PPI Contributions:
YORKSURe: The PPI contributors played a key role in shaping the reciprocal design and carefully wording explanatory materials to convey the complex information to potential participants.
NHS-Galleri: PPI work focused on the data use and sharing arrangements. Understanding patient and public concerns around use of centralised personal healthcare data and international transfer of anonymised personal data helped us design participant information materials that were commended by the ethics committee and Sponsor.
YouScreen: Collaborating with Live Through This (LGBTIQ+ cancer group) we revised trail materials to be more inclusive for transmen. Additionally, the YouScreen website featured speak aloud translations (130 languages) and provided fully translated participant information sheets (PIS) in the top five languages for each London borough.
New Paragraph
